The nuclear membrane is interesting, to say the least, the perinuclear space the “literal outside” and the two bounding membranes fluid and changing positions in an active and rarely appreciated way. I can remember in the late 1960s working at a place called “Pasadena Foundation for Medical Research” which no longer exists, watching some of the first and best cellular time lapse photography, the nuclei spinning and jerking around inside the cell, mitochondria dancing close, touching the outer nuclear membrane (in what I called nuclear pore-mitochondrial interactions in some paper long ago.
There are dozens of important proteins as part of the inner nuclear membrane, organizing and facilitating every aspect of transcription, translation, expression, silencing, condensing, organizing, modifying nuclear DNA and RNA and all components therein the likes of which I know practically nothing. I do know the organization of the inner nuclear membrane visually…. and this little segment of proteins – slightly larger it seems, than a ribosome (or about 40nm) is special! It is pseudocolored magenta, and it has ordered and deliberate spacing not seen normally (check out the rest of the inner nuclear membrane adjacent to this stretch). The nucleus itself is colored blue, and the perinuclear space, light blue, ribosomes in the cytoplasm are pseudocolored bright red-orange, the mitochondrion is orange and the cytoplasm is pale pink. Top image is pseudocolored, but the bottom image is black and white, unretouched from the original micrograph (canine, 5 yr old, untreated, alveolar type II cell, lung)
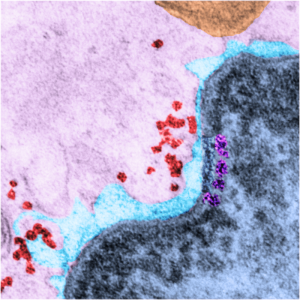
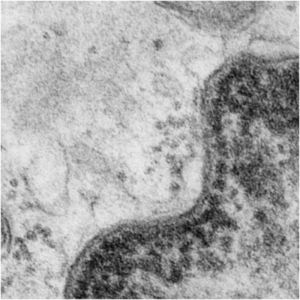 WIKIPEDIA says “There are about 60 INM (inner nuclear membrane) proteins, most of which are poorly characterized with respect to structure and function.[2] Among the few well-characterized INM proteins are lamin B receptor (LBR), the lamina associated polypeptides (LAP1, LAP2), emerin and MAN1″ italics are mine, sentence shortened as well.
WIKIPEDIA says “There are about 60 INM (inner nuclear membrane) proteins, most of which are poorly characterized with respect to structure and function.[2] Among the few well-characterized INM proteins are lamin B receptor (LBR), the lamina associated polypeptides (LAP1, LAP2), emerin and MAN1″ italics are mine, sentence shortened as well.
HERE is a link to a nice diagram and article of the inner nuclear membrane in terms of the currently known proteins. http://www.nature.com/nrm/journal/v8/n5/box/nrm2165_BX1.html