Beginning with a general truism – form follows function (a 20th century design mantra coined by Louis Sullivan originally as “form ever follows function”) is so far reaching as to make us marvel that it took so long for biologists, chemists, architects, etc, to write it down. Nature wrote that script 3,900,000,000 years ago.
To be surprised that examining the cell in detail (in this case speaking about mitochondria) is naive. It has been obvious since the invention of the electron microscope that mitochondria in different tissues look different. Before that, not much was known about these little tiny objects in the cell, or the squiggly things running around cells in culture in the early time lapse microscopy, as they are just smaller in size than the light microscope could resolve.
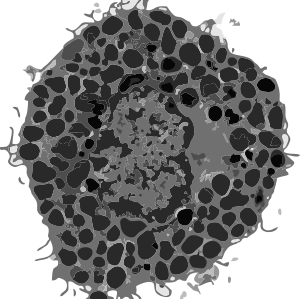
So how marvelous it is to know that when you look at a mitochondrion you are “seeing” the biological processes. Read this particular publication on the shape of cristae in mitochondria, which seems to come from a person who can “visualize the 3D as well as understand the science“. This means that of the tens of thousands of mitochondria that I have photographed that each one tells its own story, whether it was leaning toward oxidative phosphorylation, or detoxing reactive oxygen species, or moving the cell into apoptosis, or maybe preparing for mitochondrial fission or fusion, depending upon levels of stress, or getting ready to participate in autophagy. It could be moving energy out of the cell, it could be importing necessary proteins from the cytoplasm, or it could be reproducing its own distinctive mDNA, or transcribing and translating proteins on its own mitoribosomes.
So there is a glich… not all of these events show molecules that are resolvable with the electron microscopy either.
Shape (elongation or rounding) of the whole organelle, or widening of just segments of the whole organelle has meaning (visualize the bulge of a rat in a snake). The shapeso that cristae assume are clues to activities as well; whether tubular, round, triangular, with wide cristae junctions or narrow, dense matrix, numerous mitoribosomes or few, numbers of intramitochondrial granules and more that I don’t know about. The relative volume density of the matrix is also important. It is difficult to pinpoint mDNA, or even mitoribosomes… they are not easy to find like those cytoplasmic ribosomes neatly coiled on mRNA or attached to ER.
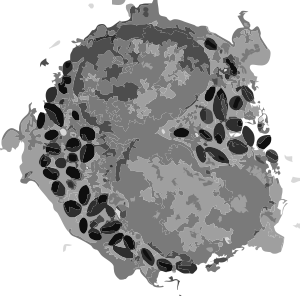
Here is a mitochondrion from which one can generalize: it is elongated (mitochondrial stress); it has narrow cristae near the outer mitochondrial membrane (hairpin bends in cristae signals lots of ATP synthase, there is an area of low cristae frequency and greater matrix, and there are some rounded cristae near the center. The cristae junctions are for the most part are quite narrow. Mitochondrion blue, ER (both smooth and rough vesicles greatly enlarged in this Gclc ko NAC rescued 50 day old mouse (#5) neg 18411 block 78375s liver mitochondrion (blue). Aside from two different types of cristae, within and elongated, and very narrow crista junctions, there are other features of this portion of an hepatocyte that are interesting, and one is the vesicle-within-a-vesicle formation of the rougn ER and also the consistent separation of ribobomes on those membranes into little groups of 7-10 ribosomes (almost like a signature) with space between the groups… (more form follows function) (orange)
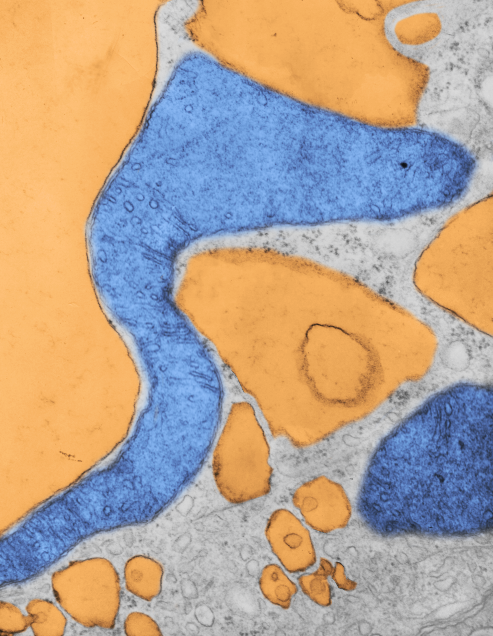
Publication above mentions that folded (tightly) cristae are working cristae, where as those that are spread apart are not functioning well. Mentioning, as i have seen in othe rpublications, that ATP synthase dimerizes or oligomerizes along the sharp curvatures of cristae. Mitochondrion above shows very tight cristae, with hairpin turns, thus one might suppose that ATP synthase is well ordered and working overtime in the Gclc ko mouse model rescued with NAC.