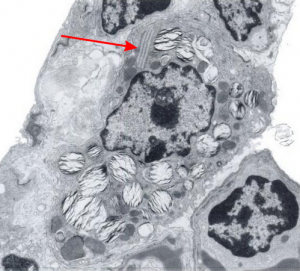
There was a group of scientists in california (I could not remember which research institute) that kindly sent me some plastic-embedded tissues of dog lung, since I had found a paper someone there that described layered protein granules in alveolar type II cells like I had seen in ferret and guinea pig and had ask them for some tissue. I could not (after 35 years, remember their names). HERE IT IS! awesome, though at least one of the researchers is deceased (J. Stara). I am taking some of their electron micrographs, cropping, editing contrast (only) and placing them next to the ones I found in guinea pig and ferret. Perfect matches. This publication is so old (1973) that there is no way that surfactant proteins could have been implicated as possible contributing factors to these granules that appeared in their ozone treated beagle dogs. The surfactant proteins had not yet been described. Ozone caused what was described (for lack of background information on overall type II cell function in those days) “metabolic alteration ….. this toxic agent”, which of course is a statement which NOW in 2016 in light of decades of research on surfactant and lung function looks patronizing.
Their RER measurements made this “approximately 754 A (75 nm) is not that far off from what was calculated by my own measurements 100nm (microscopes differ in magnification tables, and so neither is likely to be exact).
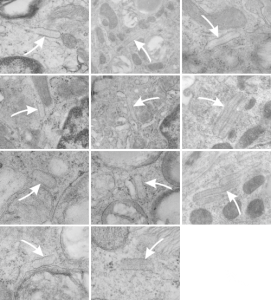
Because of their reporting of small, sometimes stacked “bar like” structures such as I have seen in untreated dogs (and looking a lot like what I have seen in rabbit type II cells). it is likely that an “excessive” amount of surfactant protein is produced and maybe stored in a multi-meric configuration) within the RER awaiting utilization in assembly within lamellar bodies or multivesicular structures. Stevens et al, 1973, mention (yippie) that there is a dense line which runs parallel to the length of the RER in these infrequently seen structures.
Ribosomes in the beagle exposed to ozone that border the RER profiles on the growing ends of the layered protein do look to be in similar positions with regard to banding of the granule to those found in ferret. My inclination is to think that there is a ribosomes on either side of the central band in each period (beagle on the left, ferret on the right – where central band is not really visible in this view) and a ribosome at the point of the darkest band (counted on two sides if there is only one period but counted only one one side if there are multiple periods) making each periodicity have three ribosomes, not counting the dark band of the adjacent period. One on a dark outside band, two on either side of the inner lighter central band. In the figure below, see comparative diagrams. the red lines with black arrows and text indicate what they think is 75 nm and I think is 100 nm (right) (give or take). Ribosomes are red, distance arrows are black, inset is enlarged from white box on bottom electron micrograph, dog on left (from Stephens et al, 1973, top image of figure 3, used without permission), ferret on right, my studies. Ribosomes are the bar marker (25-30 nm each). It is important to note that not all RER cisternae in their exposed animals showed periodicity…. just like those in guinea pigs… which I attribute to tangential sectioning and multiple intersections of different “growing ends”, and also the sudden transition from flattened cisternae of RER to the layered granule.

 More info on alveolar type II cells from Ochs, M: In humans, a single type II cell contains about 200-500 lamellar bodies, whereas in mice one finds about 50-100 lamellar bodies per type II cell.
More info on alveolar type II cells from Ochs, M: In humans, a single type II cell contains about 200-500 lamellar bodies, whereas in mice one finds about 50-100 lamellar bodies per type II cell.