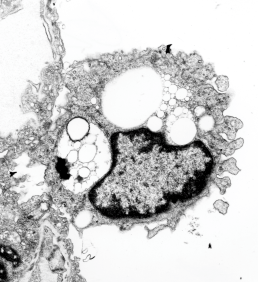
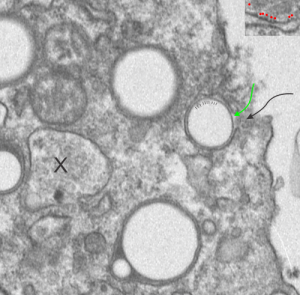
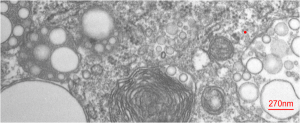
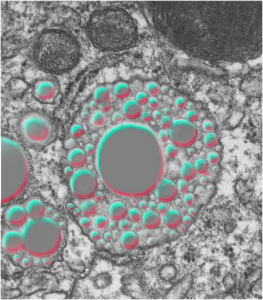
Before you pitch your old electron microscope -consider these observations.
Firstly, the human brain is a marvelous (and anticipatory) machine. You can scan at break neck speed past grid bars and open space (as one has to do when one is doing electron microscopy on the lung when it has been fixed by inflation) and the section goes by in focus. That is an amazing computational skill. If you want to waste time, scan your grid in a newer digital image scope (I used an Hitachi this morning) and i wasted half my scope time waiting for the monitor to update the image so i could tell what i just passed. Scanning by space and grid bars to find what I wanted to look at closely, and the computer is so slow at updating the images, made me realize that waiting for smear after smear of grey blur took up an inordinate amount of time andleft me feeling like i would have been better off with my old binocular viewport and my old seimens 1A microscope.
Secondly, while the function of automatic bright-dark adjustment is a nice though… again, it is very very annoying. My eyes and brain don’t need to make those contrast changes, they are automatic, and instantaneous. The lighter tissue next to a black grid bar means manual adjustment there, and the second you move more central in the grid space, it changes again. Not to reiterate the obvious, but the brain does a tremendous job of adjusting, and filtering, and adapting….. this scope didn’t even come close.
Digital image procurement on transmission electron microscopes (at least this Hitachi at children’s hospital) is a big time gobbler, and is still very very promitive. and certainly as viewed on the monitor hold no candle to film.
Thirdly, time, how much wasted time. This was supposed to be a time saver, but it recall it took about 14 small steps to capture an image on film with the Elmiskop 1A, but in fact it was less than a few seconds to perform all tasks, on this Hitachi I wait for the final image, i wait for the menu to save comes up, i hunt for the keys in the dark to label (or not label) the image, I wait for the screen to refresh. It took me less time to write the grid and block numbers in a log book and snap the picture on my antique electron microscopy.
I remember during one “annual review” when an unnamed director was in charge and he mentioned to me that i wasn’t doing enough to get new equipment…. he gave me a bad annual review…. i think because i told him it wasn’t the scope that limited microscopists, it was they 2$ eyes. I like technology…. but not for the sake of technology. I like technology when it is an improvement.
I will take the old baby on the left any day of the week.