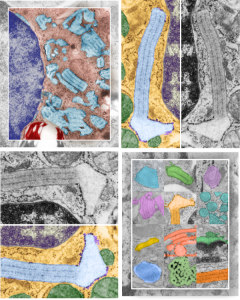
Four cover submissions for the SP-A manuscript: love the colored electron micrographs of granules. I am submitting this to Anatomical Record. They request a cover submission at the same time as initial manuscript submission, apparently, at least those are the instructions. Typically I think the manuscript has to be accepted before cover submissions are made. Oh well, I am just hoping for the best. Preview here.
Category Archives: Layered intracisternal protein granules in mammalian lung
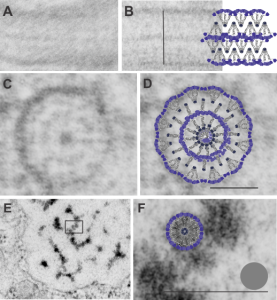
3 diagrams as putative fits for alveolar type II cell granule
So tired of looking at this structure and trying to brainstorm about how things fit together and whether SP-A is a good protein fit for this very regular and very nice molecular alignment within the RER of alveolar type II cells of several species.
A. Single period (RER membrane on top and bottom) from ferret show outer dense layers and less dense central layer. B is also a single period of a granule from guinea pig which is not bounded by RER but by another period above and below. Less electron-density accompanies of outer dense layers occurs when adjacent periods are present. C is an end on view of a cylindrical granule from guinea pig, almost 200 nm in diameter which could be twice the single period width. The inner dense central layer would be compressed into the central density. The RER membranes are seen on the upper and left on the outer edges, and periodicities for both the less dense center layers and dense layers appear as concentric rings. D, is the same image as C, with a cylindrical model of an end-on view of the possible arrangement of SP-A molecules which could account for this structure. The number of densities in the concentric layers was used to calculate how many molecules to use for the model. Vertical lines (shown in Fig. 5 as well) became radial spokes. Bars = 100 nm. E. On rare occasions, and only in guinea pig, round and dense fuzzy-ball like structures were seen squeezed between the typical 100 nm periods. Box in E was enlarged in F to show the relative sizes of the round densities. A diagram of a potential SP-A fuzzy ball, approximately 30-40 nm in diameter approximates measurements of the spherical densities. Grey circle is ribosome size for comparison. Bars = 100 nm
Diagram: SP-A and lamellar bodies
An article by Palaniyar N, Ikegami M, Korfhagen T, Whitsett J, McCormack FX. 2001 entitled Domains of surfactant protein A that affect protein oligomerization, lipid structure and surface tension, published in Comparative Biochemistry and Physiology 129:109-127, presents a diagram of how the 18-mer of SP-A might integrate itself into the outer lamellae of lamellar bodies in alveolar type II cells. I did notice an inconsistency, perhaps, in the diagrams. The bottom diagram (left bottom) shows the SP-A molecules with their N terminals all pointing inside, or towards each other in the center of these tubular myelin structures (the diagram is a 2D model of a 3D structure, hence “tubular”). In the granule found in alveolar type II cells which I have been trying to describe morphologically for several years has a layering pattern that suggests that the carbohydrate recognition domains are actually pointing “away” from each other. This is replicated in the diagram to the left. The inconsistency comes in how the SP-A molecules are represented in the top portion of the diagrams, which I duplicated in part, and re-oriented the SP-A molecules to match the tubular myelin arrangement shown in their figure (bottom left). To me this makes more sense, and it also is consistent with the layering of the alveolar type II cell granule in guinea pit, ferret, and dog, and in addition, with the outward orientation proposed for the Birbeck granule (the C-type lectin, langerin). Comments are welcome. My edits to their diagram are in the red dotted inset to right.
Alveolar type II cell of a cat: surfactant granule?
This electron micrograph is from a cat lung, and is an alveolar type II cell. There are some areas of dilated RER which seem to fit the description of a surfactant protein A type organized granule. In particular this banding, which is admittedly faint (not section chatter as just above it there is the membrane of a mitochondrion which is pretty much in line with the middle band of this granule which might make one think the banding is chatter, but the dense line does not extent further into the micrograph past the rounded edge of the mitochonrion, therefore is not likely to be chatter.
I think it is the best example of layering (oligomerization of the protein) within the RER that I have of the few electron micrographs of cat lung that I have.
A lamellar body is within the image on the left of the nucleus in the lower magnification view. The enlarged portion is demarked with a white box (in the left hand image). Judge size by using the two ribosomes on the upper membrane of the perinuclear area that has the layering (layers are pointed out with black arrows). Ribosomes would be about 27 nm in diameter, making the banding about four ribosomes wide…. or more or less equivalent to the 100 nm layering predicted to be SP-A. Yep, circumstantial, but quite likely.
 Cat number 3, alveolar type II cell electron micrograph, neg 7268 block 24564, original magnification 5000 x, enlarged 4x.
Cat number 3, alveolar type II cell electron micrograph, neg 7268 block 24564, original magnification 5000 x, enlarged 4x.
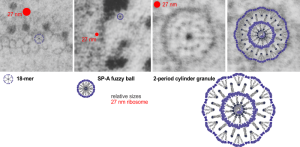
SP-A: three possible configurations to match electron microscopy of alveolar type II cell granules
Still working on this granule in alveolar type II cells. I think there are three levels of organization to the cylindrical and circumferential manifestations of whatever surfactant protein comprises these granules. The original 18-mer, well confirmed by numerous other scientists, on the left, relative to the size of a ribosome, and also with the hexagonal order seen in tangential sections of the outer dense layer of the granule. Middle image is the proposed SP-A fuzzy ball which seems to be a spherical organization of the same 18-mer, just with something different happening in the center region of the sphere. I can’t even guess what that organization (missing perhaps some of the CRD regions of SP-A?) to make things fit. This image also relative to the size of a single ribosome (red dot). Then on the right hand side is a double-layered cylindrical granule (this one likely from guinea pig) which has a diameter of about 200 nm, which means is likely has a column of four molecules for each radius. Size of the actual superimposed cylindrical period, and then a relatively sized – diagram… sized relative to the other spherical or concentric configurations of SP-A. The micrographs do pretty much dictate the arrangements, or at least that was what dictated the arrangements of the SP-A stick figure I used (which was vectorized from models of SP-A in publications.
A better fit model for langerin and profiles of Birbeck granules
Lots of images have been published for Burbeck granules. I am interested in them only because they seem to be the only other C-type lectin which has a substructure that is organized and obvious when viewed with electron microscopy. Well not the only, because I think that SP-D has fuzzy ball structure that is seen with TEM. It is puzzling why MBP and some other lectins don’t assemble into orderly granules like langerin, and SP-A (at least I think it is SP-A).
So here is a composite that I have made from TEMs posted everywhere in the literature, lined up with the 50 nm distance between the ER membranes, and such a very predictable substructure of proteins organized within. One publication in Plos ONE (Glycosaminoglycans Are Interactants of Langerin: Comparison with gp120 Highlights an Unexpected Calcium-Independent Binding Mode. Eric Chabrol, Alessandra Nurisso, Antoine Daina, Emilie Vassal-Stermann, Michel Thepaut, Eric Girard, Romain R. Vivès, Franck Fieschi) gives a molecular structure and a hypothetical overlay onto TEM images. I think they missed it by a little…. for starters their intramembrane part of the molecule is NOT shown in their diagrams, which makes the molecular fit difficult since the CRD of the molecule are a relatively larger portion of the actual TEMs than is seen in their molecular diagram (line diagrams on the far right of the image). Additionally, there is a cytoplasmic portion of langerin, at opposite ends of the mirrored CRD portions of langerin which their diagram does not show (don’t know why they chose not to show it).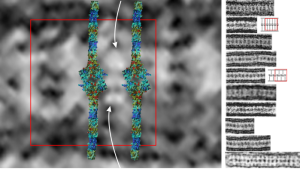 I vectorized their diagram and have placed it back in the 50 nm membrane boundaries of the ER with the transmembrane portions actually existing and that makes the size ration of the model molecule fit the actual TEM much better.
I vectorized their diagram and have placed it back in the 50 nm membrane boundaries of the ER with the transmembrane portions actually existing and that makes the size ration of the model molecule fit the actual TEM much better.
Again, my only interest in this is that I am trying to ascertain whether my thought that SP-A is responsible for the granule in guinea pig and ferret type II alveolar cells true.
Right side of the diagram is a collection of Birbeck granule TEMs sized to 50 nm, the image on the right shows a red 50 nm bounding box, and four langerin molecules oriented CRD inward, and transmembrane areas through the ER membrane. Just my opinion here but there is another molecule or some portion of langerin which is “between” the four diagrams superimposed. White arrows point there. Their diagrams (two alternatives) are shown in red and grey to the right of the actual TEMs of Birbeck granules.
Fuzzy ball vs ribosome
Here is a comparison, a diagram of what might be an SP-A fuzzy ball at something like 47-50 nm, and a nearby ribosome @27 or so nm. The image on left, shows inset box from the granule in this alveolar type II cell from which the inset is derived. My diagram isn’t exact, but it might be close. These images are from guinea pig (now famous) 301.
Are these fuzzy balls surfactant protein A or SP-D?
There is one guinea pig in this group of animals that was completely different, in the sense that all the surfactant granules within alveolar type II cells in this animal are just overpowering all other cytoplasmic elements. This was a control animal which makes for interesting speculation, maybe even an oblique reference to the fact that this structure is a response to environmental influence (in this case probably a massive pulmonary infection or immune disorder. At least there was no experimental reason for alveolar type II cells to display so many granules.
Posted previously are images of an interesting variation on the granules, those which, in between the 100 nm layering pattern that is most prominent, some fuzzy rounded ball-like groups of protein exist. A previous post that mentions these structures, in that case they were given a dimension of 50-60 nm… here I measured them more like 49 nm… so basically the same. I was wondering at that time if they were surfactant protein A or not. I am wondering now if they might be surfactant protein D. Well, still leaning toward SP-A, in a configuration that would be two molecules “deep” or half the width of the banding seen in the granules.
I did find this reference from Crouch et al, 1994 which had rat SP-D images that were awesome, however the dimensions don’t quite fit… and they give 46 nm as the size for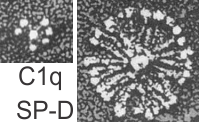 a single arm of the SP-D molecule, and that is exactly half of what I measure as the full diameter of one of these dense structures diameter (47 nm). They calibrated their microscope well, I am not arguing with their final dimensions. From their paper are two screen shots C1q (left) and SP-D (right). The magnification is not given in a micron marker bar, but C1q is just a little smaller than SP-A, there fore if the relative magnifications are the same in these two, then that makes SP-D about twice as big as SP-A would be so maybe 50 microns which sort of contradicts the size given. If C1q has a spread of the CRD close to SP-A (which is 20-20 nm) and picture on left is 20-30 nm diameter, then it appears that the SP-D here is about 2.5 times larger than SP-A would be and that is more like 70 nm or even 100 nm, and a previous post on this blog calculated the round SP-D at 100
a single arm of the SP-D molecule, and that is exactly half of what I measure as the full diameter of one of these dense structures diameter (47 nm). They calibrated their microscope well, I am not arguing with their final dimensions. From their paper are two screen shots C1q (left) and SP-D (right). The magnification is not given in a micron marker bar, but C1q is just a little smaller than SP-A, there fore if the relative magnifications are the same in these two, then that makes SP-D about twice as big as SP-A would be so maybe 50 microns which sort of contradicts the size given. If C1q has a spread of the CRD close to SP-A (which is 20-20 nm) and picture on left is 20-30 nm diameter, then it appears that the SP-D here is about 2.5 times larger than SP-A would be and that is more like 70 nm or even 100 nm, and a previous post on this blog calculated the round SP-D at 100  nm. I am opting for the fuzzy balls in the granules of alveolar type II cells in this study to be SP-A. Per diagram.
nm. I am opting for the fuzzy balls in the granules of alveolar type II cells in this study to be SP-A. Per diagram.
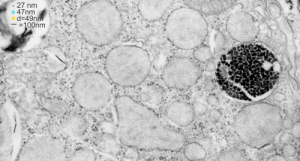
The image below (9124_17082_gpig_301_alveolar type II cell with granules, lamellar body (upper left under the legend) and a fuzzy all inclusion (in the RER i believe). I saw these fuzzy balls embedded within granules but this is the only time I saw a cluster of them as a separate granule. Image below show the fuzzy balls clustered on the right, the figure below that shows relative sizes of ribosomes (red dots of 27 nm) and some examples of the diameter of the fuzzy balls at something around 47 nm and an optimally sectioned fuzzy ball (greatest diameter) of almost 50 nm.
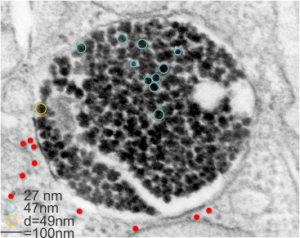
These fuzzy balls can occur, per above comments, within the granules which are otherwise typically without these spherical densities. micrograph 9864_17085_gpig_301. The red dots on right are more or less equivalent to 27 nm diameter (ribosome size) and the red bar is therefore equivalent to 100 nm. Because of the lack of prominent layering and the added width (red bar) for the periods, it is presumed that this represents a tangential cut, and the size of the electron dense round balls (blue circles in images on right) is something around 47 nm (which is similar to estimates in other micrographs based on the size of a ribosome. So these fuzzy balls can be seen both as separate membrane bound entities or found between the period layers of granules. Unretouched photo on the left, photo on the right has the end-on single circular profiles highlighted…. looking like bulls-eyes. 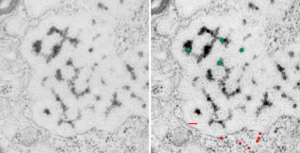
What do fixation and staining do
Methods of fixation and staining allow ultrastructural components of each cell type to be preserved and resolved differently. Glutaraldehyde based, non-coagulant-fixatives encourage direct cross-links with amines and other nucleophiles, most notably lysine and arginine, and cause deformation of the alpha-helix structures in proteins. Paraformaldehyde cross-links basic amino acid lysine, as well. Osmium tetroxide, in these studies was always used as a secondary fixative, except for beagle dog, where it was used as a primary fixative, helps to preserve lipids, and cross-links at unsaturated sites in membrane lipids and nucleophiles like amino and sulfhydryl groups. Osmium tetroxide enhances overall electron density of the specimen, adding contrast. Contrast is further enhanced by positive staining with heavy metal salts (uranyl acetate, enhancing lipids, nucleoproteins and proteins) and lead citrate (enhancing the contrast of proteins, membranes and glycogen) (Frankl, 2015; wikipedia). These all fit well with the type of layering (electron dense banding) found in alveolar type II cell granules.
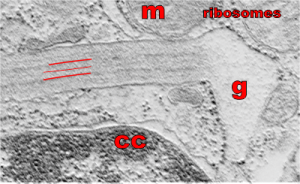
So what electron density in the structure of the alveolar type II cell reveal about the molecular structure of the layered protein. Well, surfactant protein A (and D also) has helical regions, probably hopefully the less dense central layers have a lot of lysine and sulfhydryl groups. This might fit well with surfactant protein A patterning. Three red lines = 2 outer dense layers and less dense central layer; m=mitochondrion; ribosomes are the black dots (nucleoprotein staining with uranyl acetate), especially situated on the electron lucent racket end of the granule; cc= condensed nuclear chromatin (nucleoprotein staining with uranyl acetate), in the rounded portion of nucleus in the lower left corner of the micrograph. This image was shadowed with an exact replica, 68% transparency, and a few pixels to right and lower than bottom image. one of my favorite electron micrographs of these alveolar type II cell granules .
Does alveolar type II cell granule exit at the apical plasmalemma?
The highly ordered protein granule in the alveolar type II cell of some species doesn’t not often show any signs of where it “goes” in the cytoplasm, whether to exit at the apical plasmalemma or whether only to be dumped into the lamellar body to exit the apical plasmalemma with immature surfactant that the lamellar body holds. This is probably an image of the closest encounter I have seen between a granule and the apical membrane, and it is not complete, nor definitive at all, but just a reminder of the fact that the issue remains unresolved. See here four granules, the one on the left is very close to the apical membrane. There is a white line around a nearby apical membrane fold (typically called a microvillus, but certainly not a rigid one) and the bracket denotes the space between the former and the latter — close at best, but not a membrane fusion and release.
Banding is clearly evident within the granule. Sadly, the back of this micrograph is labeled “duplicate” but i cannot find the original with the description of negative number, block number animal number and species, but it will be either guinea pig or ferret.
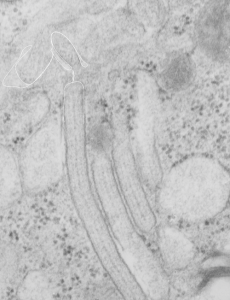 Just for comparison, here is a close encounter of granules with a lamellar body, which is actually a fairly frequent event. Lamellar bodies on left upper and lower right. three angled structures are granules (you can see the linear banding in the center one.
Just for comparison, here is a close encounter of granules with a lamellar body, which is actually a fairly frequent event. Lamellar bodies on left upper and lower right. three angled structures are granules (you can see the linear banding in the center one.