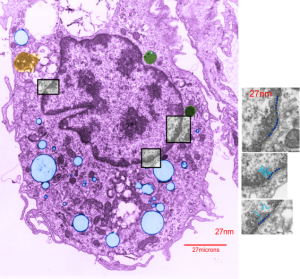
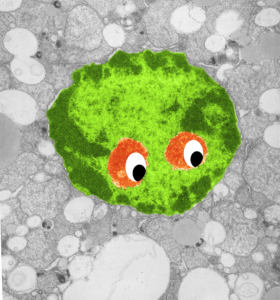
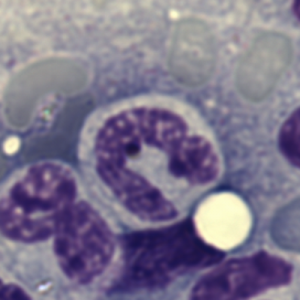
Here is an electron micrograph of an alveolar macrophage from a mouse that liquid-breathed E2 for 3 hours and allowed to recover for 17 days. The droplets of E2 (which i pseudocolored blue) are well defined and surrounded by a relatively prominent membrane and likely have some characteristics of lysosomes or endosomes because of the peripheral electron density. Two multivesicular bodies/late endosome type structures (pseudocolored green) show tiny bits of E2 (blue) as well (at least this is how i interpret it). This was sort of typical for E2 droplets in similarly treated mice. Organelles within this alveolar macrophage seem relatively healthy and importantly, the condensed chromatin is typical of non-activated macrophages in general. There is some endocytic function remaining as there is an inclusion of tubular myelin (orange) contrasted with the pink nucleus and cytoplasm.
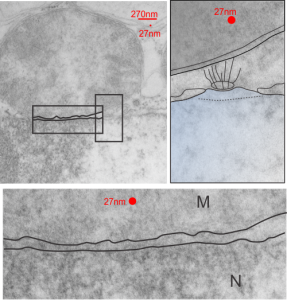
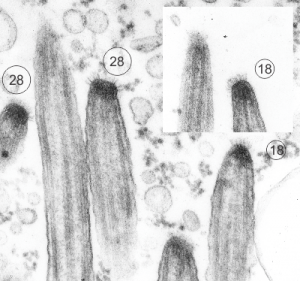
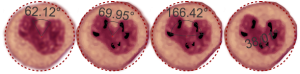
The three grey insets (notice that the bottom one has been mirrored horizontally) show a distinct periodicity to the most peripheral chromatin against the inner nuclear membrane. And there are places where the size and spatial arrangement of the densities change (smaller “somes” – whatever state this condensed chromatin is in with its attendant RNA and other proteins). The spacing (a little smaller than the dark blue dots) is something close to 30nm when I use a mean diameter of a cytoplasmic ribosome as the measure of 27nm for this image. 1432_mouse_lung_liquid_breathing_3hr_17d_recovery.