I worked hard and lobbied continuously for this cover. Here is a link to the journal in general.
Monthly Archives: September 2017
Substructure of the lysosomes containing perfluorodecyl iodide
Substructure of the lysosomes containing perfluorodecyl iodide is very interesting. There are issues both with the lysosomal membrane, which in some places may be continuous with other membranes, in particular, i think maybe the RER. Because perfluorocemicals are kind of “slippery” and I think they can travel easily retrograde-style back up into the RER, then it makes sense that some profiles of RER would be seen at sites within the lysosome periphery and the adjacent cytoplasm. I haven’t been able to equivocally find ribosomes on the actual lysosomal membrane to verify that concept. There are perfluorodecyl iodide tiny crystals…. this is interesting…. an obvious type of fracture or separation plane along the long axis of the larger crystal inclusion certainly is evident…. and i am more convinced that the pattern of lysosomes (a dotted or tubular pattern) is on the order of 20-30nm diameter or width.
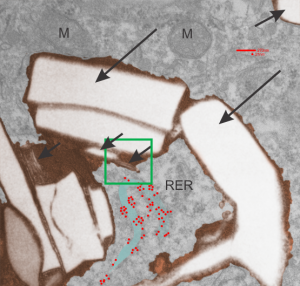
Here is an electron micrograph: mouse, neg 9722_10%IPFD, 5%F68 infused 100cc/kg, 8-14-73 sac 5-8-74, 267 days post infusion; liver. Brown areas=lysosomal enzymes within a bounding membrane, white=IPFD, red dots are ribosomes; pale green is cisterna of RER, green box is area enlarged below.
So the bottom two images, left unretouched, right unretouched but with translucent overlays where I see a pattern within the lysosomal enzyme membrane. Arrows here point to fractured off crystals of IPFD, and the red line is a marker for 27nm which is the same size as the ribosome measured (top inset)…. this makes me wonder if there is some connection between this pattern and lysosomal contents.
Periodicity at ends of IPFD crystals in macrophages
I looks like the lysosomal enzymes at the end of the IPFD crystal inclusion-structures might be tubular. But then as the enzymes pass away from the IPFD itself, there can be a coiling up of the protein with less rigidity. Measurements at ends of IPFD look smaller than those in the lysosom proper… may be error of just having only measured a single crystal. Same negative and block as previous post, and the ends of the IPFD crystal are colored bluish, and the area measure for ribosome size is in red. Enlargements below, with areas specific measured highlighted. The dense part of the period and the lucent areas beside it.
Texture comparisons: background, cytoplasm and IPFD-lysosome
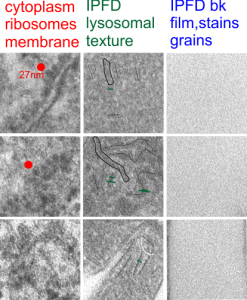
I am trying to determine whether there is a predictable texture to the proteins within phagolysosomes which contain IPFD (perfluorodecyl iodide)(which I am positive there is–so this is academic). I have compared territories from one micrograph with marked inset boxes: red boxes=cytoplasm with a ribosome size marker for comparisons of texture-size); green boxes=portions of the compact and textured lysosomal proteins found just within the limiting membrane of the phagolysosomes which contain IPFD; blue boxes=the background texture of the micrograph found over the “empty” IPFD granules which consists of the acetate film grain and the stains– osmium, uranyl acetate and lead citrate grains. No photographic paper grains are present as this was scanned at 5000 ppi from the negative.
I measured a similar texture pattern from the end-on portion of the lysosomal proteins in last post, and dimensions are similar but not identical… likely my fault, not the fault of the proteins. So todays measurements are something around the following: dense areas at around 17nm (corresponding to the “spot” in yesterdays post which was around 13nm) and the surrounding more electron lucent space measures about 34nm (which in yesterdays post was closer to 40nm)–all likely resulting from variations in how i measured ribosome size, which is the benchmark.
Image below is from a randomly selected phagocyte in the liver (I cannot identify whether this is a Kupffer cell or other phagocytic cell type). These cells can be closely aligned with sinusoids but also near portal tetrads. They are typically “overflowing” with IPFD particles. IPFD stays around for months, unlike other some perfluorochemicals.
Neg 9722 block 3775 10% IPFD 5%F68 injected 8 14 1973 100cc/kg sac 5 8 1974 mouse liver 267 days.
IPFD crystals in lysosomes
RedI cut around the top end of the crystal inclusion in the image from the previous post, and erased (in photoshop) areas that allowed me to pseudocolor the objects that I could see in the lysosomal phase of this phagososome. I think the periodicity is going to be interesting with a 13-15nm round central dense area and a lucent area around that density (like a donut), still with a layer of dense protein or a trilaminar membrane.
It was fairly easy to see the repeating dots and lucent surroundings emerge as a repeating pattern along the terminal membrane at the long axis of the IPFD inclusion. IPFD crystals show for the most part a definite polarity, and any substructure in the IPFD inclusion itself is see on the butt ends of the short width axis, while a very smooth bounding area is seen on both sides running in a parallel direction.
Below, same pix as 9 13 2017 post but with a bounding box which aligns to the inset image below again.
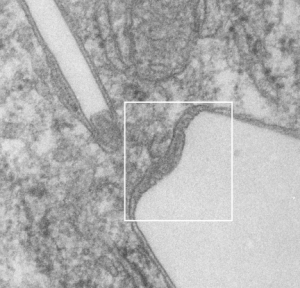
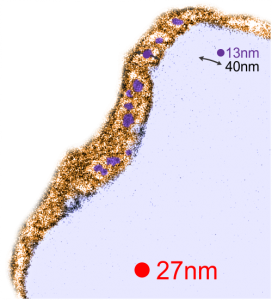
Red dot=ribosome at 27nm diameter, purple dot=central densities, tangential end sections, 40nm dimension for a cross sectional diameter of the electron lucent area surrounding the purple dot, and 13 nm dimension is for the purple dots (some larger some smaller).
Awesome IPFD crystals in lysosomes
Perfluorodecyl iodide was examined once upon a long time ago as a possible PFC for blood substitutes. There are a couple of tissue blocks which I am re-examining to see whether there is a periodicity to the lysosomes that are included in these amazing phago-lysosomal structures, left over footprints as it were, to the presence of IPFD in macrophages (Kupffer cells, fat storing cells?, circulating macrophages, in the liver).
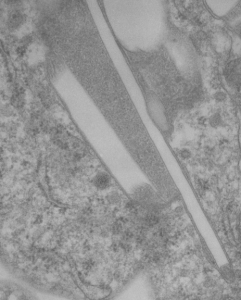
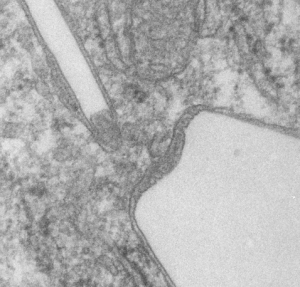
This lower micrograph has very definite structure at the ends (one top one bottom) of the crystals. It is a little unfortunate that at the time i printed these negatives (back in the ‘wet darkroom days’ that I didnt use a finer grade paper and developer. Too much grain, but the little indents and rounded areas with a punctate density are going to be relly interesting. I think the lucent areas beside each will measure out the same as some fine lines that I see at the ends of other crystals.
Comparing perfluorocarbon emulsions: Clark et al, 1974
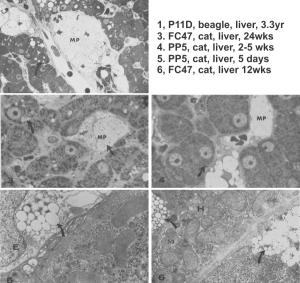
I ran into this publication that I had not even put in my own folder of pdfs, that was published back in 1974, Clark LC, JR, et al, in Microvascular Research. I was sort of surprised to see how much effort went into the assessment of perfluorocarbon blood substitute emulsion particles in the livers of several cats receiving either 1) no emulsion, 2) PP5 emulsion or, 3) FC47. Early on it was evident to me that the re-emulsification of fluorocarbons in vivo was quite dependent upon the chemical characteristics (but i dont know which) of the PFC, combined with — or prompting the lysosomal response, coupled with the apparent diffusion and aging characteristics of particles within phagolysosomes.
I have extracted the micrographs from this publication and added the descriptions (which were unfortunately difficult to find) from the publication which shows that P11D and FC47 clearly differ from PP5 in the responses they elicit from the phagocytes in the liver (likely Kupffer cells and fat-storing cells, are RE cells. Top three images are light micrographs, bottom two images are transmission electron micrographs. No bar markers unfortunately but in the top three use a nuclear profile as about 15 microns, in the bottom two, use the short diameter of a mitochondrion as about 0.2 microns.
 In this publication Clark notes that PFC ring compounds with O, and N, and straight chain PFC with O, hang around for a more extended time than those with just C and F.
In this publication Clark notes that PFC ring compounds with O, and N, and straight chain PFC with O, hang around for a more extended time than those with just C and F.
Do perfluorocarbon emulsions nucleate while in blood vessels?
Do perfluorocarbon emulsions nucleate while in blood vessels? Seems likely, particularly since there are shear forces in circulation that are brought to bear and these are variable, depending upon blood volume, blood pressure, and percent of hemodilution during the procedure and thousands of other things that are likely going to be brought to bear.
Thoughts on perfluorocarbon droplets: coalescence or diffusion
It is an obvious thought, but not one I considered, but if one gives perfluorocarbon based blood substitute emulsions to individuals, their body temperature may change the manner of(diffusion and/or coalescence of droplets), shear forces in the blood stream, and rate, and morphology of droplets once engulfed (large droplets, smaller droplets, rate and amount of re-emulsification of the perfluorocarbons within lysosome/phagolysosomes). So what happens if someones body temperature is 97.5 or what if the recipient of artificial blood is febrile at 104? This concept was brought up in my mind by a report, a publication by Mara G. Freire et al, 2005 J. Colloid and Interface Science, of behavior of droplets in experiments in vitro. I believe that when we did infusions of perfluorocarbon emulsions in larger mammals (the Leland Clark Jr, laboratory, back in the 1970s) body temperature was always recorded, however I don’t believe those numbers were part of the morphometric metadata for “mean particle size” of droplets within lysosomes, in vivo, in measurements of particle size in liver biopsy samples.
Two areas of emulsion stability are relevant (at least two) in vivo: within the blood stream and within the cell. The latter looks to have two processes going on: — aging of the emulsion which includes coalescence of droplets. Freire et al 2005 indicate that droplet size is an indicator of emulsion age (i would agree in the blood stream), but with within the cell, perhaps there is a critical size – as a divide, where at one size and < that the act of droplets growing smaller to be lost by diffusion predominates, and at that critical size or larger –oswald ripening or coalescence occurs. The “divide” as it were, may have something to do with the 1) physical and chemical characteristics of the perfluorochemical, 2) the enzymes poured into phagolysosomes — likely to vary with cell type, temperature, state of enzyme activation from baseline, and in response to the perfluorochemical, 3) proximity of an “air” interface (as in the lung), 4) mechanical forces (as in inflation, deflation of the lobes of the lung or flow through the blood vessels). Whether hydrate nucleation occurs?…. would be nice to find some literature sort this out.
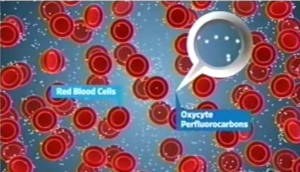
So in looking for microscopy and perfluorocarbon emulsions I ran onto a cute diagram, though probably so far from reality as to be laughable, and perhaps only relevant for the first nanosecond before the white cells begin gulping emulsion particles up, and before nucleation or coalescing of the emulsion particles begins to occur in the blood stream. I don’t know to whom the credit goes, whether to the writer of his thesis, Cameron Smith, Virginia Commonwealth University (wherein I found the cartoon) or to Oxycyte, referenced in his legend, here is –A cartoon illustrating the relative size of red blood cells (diameter approx 6-8 µm) compared to Oxycyte® PFC emulsion particle size (approx 0.1-04µm).
Cutest typos ever – colonary
I should be gracious, as it is common for me to make typos just the same as everyone else, but sometimes i find one that just tickles me. This, in a book whose editor is japanese, just was so cute i had to list it. The “L” “R” thing is quite evident. And while this is a post about a typo, i still give praise to anyone who is fluent in more than one language, power to them. This is just for a smile. Being an anatomist, i wondered if somehow i had missed this special type of arterial disease of the colon; I did google the word… nope, google thought it should be “coronary” as well.