 Wikipedia’s post on “all about fluorine’– this element is so unique, and with fluorine man has made some wacko new compounds. There is only a small post on perfluorinated blood substitutes down at the bottom. I was not amused by the fact that there were “fluorine martyrs” to fluorine chemistry experiments gone bad. And in the old days of science, those guys really took their work seriously, without understanding (or knowing) consequences, and their lives in their hands. I wonder if technology outpaces common sense. Of course I don’t wonder. ha ha… it does, all the time, every day.
Wikipedia’s post on “all about fluorine’– this element is so unique, and with fluorine man has made some wacko new compounds. There is only a small post on perfluorinated blood substitutes down at the bottom. I was not amused by the fact that there were “fluorine martyrs” to fluorine chemistry experiments gone bad. And in the old days of science, those guys really took their work seriously, without understanding (or knowing) consequences, and their lives in their hands. I wonder if technology outpaces common sense. Of course I don’t wonder. ha ha… it does, all the time, every day.
Monthly Archives: September 2017
Inner ear stereo-pair
Many years ago I was interested in reconstructing 3D images from sections of inner ear. I still have many of those slides and images. At that time my son Daniel Miller was into computer graphics. He took my highlighted 2D sections (as shown in the bottom image) and created this stereo pair. Cochelar duct yellow, ampulla of the semicircular canals white.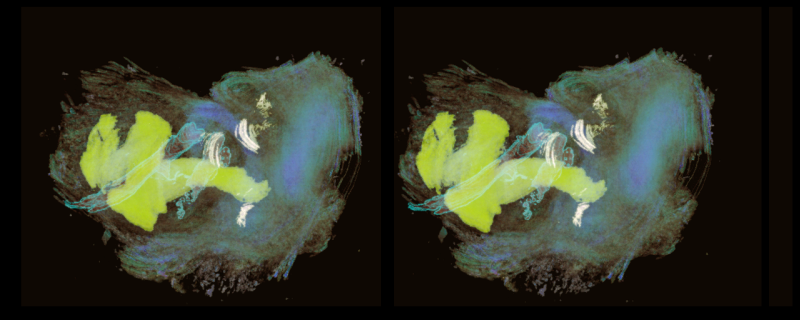
It looks like we submitted a cover illustration as well. I cant even remember if it was successful.
Perfluorodecyl iodide: a peroxisome proliferator
Going through archived images of the livers of mice which received infusions of iodo containing perfluorochemicals, I ran onto a hand-written comment which made me laugh. It was “funky print given under pressure to LC Clark (Jr) that perfluorodecyl iodide increases tremendously the number and size of microbodies (aka peroxisomes) in the mouse liver” (words in italics added today for clarity). Apparently no one wanted to hear that news in 1973 or 4… perhaps because it was a “damning” mark on the possible use of perfluorochemical formulations in general, and specifically for artificial – blood substitutes.
Wikipedia summary is here: “They are involved in catabolism of very long chain fatty acids, branched chain fatty acids, D-amino acids, and polyamines, reduction of reactive oxygen species – specifically hydrogen peroxide; – and biosynthesis of plasmalogens, such as ether phospholipids critical for the normal function of mammalian brains and lungs. They also contain approximately 10% of the total activity of two enzymes in the pentose phosphate pathway, which is important for energy metabolism.”
Below are three images, one of liver which shows the very large peroxisomes, in this particular micrograph (not all shown here) they were a volume density of about 30% of the cytoplasm which puts IPFD up there with the best of the proliferators; mitochondria for comparison were about 33% of the cytoplasm. Consensus has peroxisomes at more like 2% of the volume density of the hepatocyte under unstimulated conditions.
The story will be complicated as there are more than 30 enzymes which are in peroxisomes: and the patterning that is seen in the enzymes within the phagolysosomes containing IPFD looks to be similarly wound as the enzymes within peroxisomes in the hepatocytes (top two image groups). From the internet the middle three images (used without permission as a screen print of the insanely egotistical Dr. Jastro TEM collection who most certainly used public funds for this research and collection of micrographs and yet thinks he has ownership of these images…. not civil at all in my opinion and shame on him) is an hepatocyte peroxisome (which i measured against a nearby ribosome and also the micron bar marker, for size) shows a coarse texture, somewhat coiled. The latter and my peroxisome images (top), added from a portion of an hepatocyte from the liver which received IPFD and measured against a nearby ribosome, show similar “oligomerization” or “substructural” protein patterning.
Some protein(s) is creating this texture. It would be interesting to know if the protein in these microbodies (aka peroxisomes) is the same as that which is found in abundance in the phagolysosomes containing IPFD. Bottom micrograph is a portion of hepatocyte cytoplasm from IPFD infused mouse more than 200 days post infusion from which my (top two) images are derived.


Finding the crystal width pattern for perfluorodecyl iodide
I have felt there was a pattern to the width of the perfluorodecyl iodide crystals in the micrographs that I have for some time. Today I decided to measure just two sets of crystals from two different lysosomal structures from two different cells. The graph below shows that there might be a stepwise increase to the thickness of the crystals of something on the order of 18-20nm (or maybe half that). The data from 4-8 measurements along the wide-axis of individual crystals were measured an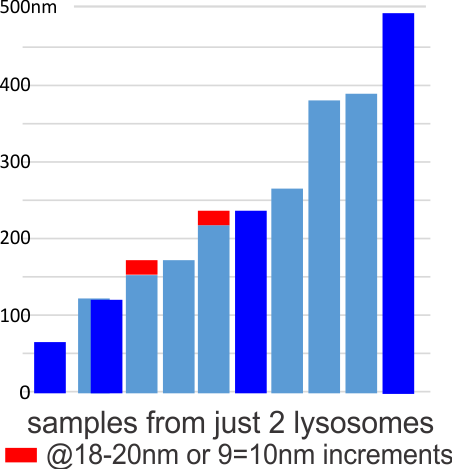 d normalized to the 27nm size of an adjacent ribosome. The mean widths sorted in ascending array and the second plot measured identically (had a few more crystals to measure) is superimposed. The red rectangles might indicate an incremental increase in size. Just thinking on this. I did find a couple of publications on “incremental pattern mining” which would be fun to examine. Hopefully I will find someone with software already in place to do this. The actual micrographs and positions of measurement are shown below. Red dots=27nm a presumed size for a mammalian ribosome, in each micrograph. Dont miss the lysosomal protein “snake-like protein with dark central stripe” mentioned in previous which is very clear in the top portion of the top electron micrograph. In this micrograph the values are 18-20 and 9-10 nm respectively for the two-band protein (similarly to previous post HERE).
d normalized to the 27nm size of an adjacent ribosome. The mean widths sorted in ascending array and the second plot measured identically (had a few more crystals to measure) is superimposed. The red rectangles might indicate an incremental increase in size. Just thinking on this. I did find a couple of publications on “incremental pattern mining” which would be fun to examine. Hopefully I will find someone with software already in place to do this. The actual micrographs and positions of measurement are shown below. Red dots=27nm a presumed size for a mammalian ribosome, in each micrograph. Dont miss the lysosomal protein “snake-like protein with dark central stripe” mentioned in previous which is very clear in the top portion of the top electron micrograph. In this micrograph the values are 18-20 and 9-10 nm respectively for the two-band protein (similarly to previous post HERE).


Texting, FB, generally not paying attention to fellow humans in real life
Who remembers “FANG”
Endosomes in phagocytes containing perfluorodecyl iodide crystals show unique substructure
The phagolysosomes in phagocytes which containing perfluorodecyl iodide crystals (in this case the perfluorocarbon has been retained in the body for more than 250 days – mouse liver) show huge amounts of lysosomal enzymes. The collection of macrophages (even multinucleate macrophages) don’t appear to initiate a huge collagen response, and inflammatory cells such as neutrophils, just are not that common, even when phagocytes are plentiful.
The ultrastructural characteristics of the lysosomal enzymes contents within phagolysosomes containing IPFD crystals can vary dramatically within the same cell and in adjacent lysosomes, and within the same lysosome. This varies from a very fine homogeneous non-textured content in some areas to areas with a more coarse fibril-like texture, appearing like a lumpy (sausage link type) strand or a twisted strand (of two separate elements, or a two strand braid). In some cases the fibrils appear to be solid on cross section, but in other cases more like a tubule. This underscores the variability that would be found in a twisted fibril, allowing for the explanation of both “hollow” and “solid” profiles on cross section.
Transition zones between the types of enzyme ultrastructure are found within the same lysosome suggesting some protein overlap or continuity. There are some areas of very dense accumulation of proteins.
IPFD crystals may be large or small, and most display some obvious directionality, usually the longer side having a thin dense protein (or trilaminar membrane?) coat, but at the ends showing considerable “striping” or dragging out, or whispy periodicity.
IPFD crystals often appear in cross section to be more rounded, or even round.
Some common characteristics of the fibrillar texture of lysosomal contents of IPFD filled lysosomes follows:
- relatively even diameter at widest and narrowest areas
- often looped and tangled, folded looking
- equally even spacing seen as a predominant lucent area surrounding each strand
- often running in parallel
- higher mag shows sub-layering
This image below shows a very prominent (yet small in terms of nm) level of periodicity. May not be the smallest since I havn’t be able to find any information on the actual crystal chemical structure of perfluorodecyl iodide), but pretty small for conventional electron microscopy to propose, but this is so obvious that it certainly warrants comment. The single space (inset is same size as lower image and placed on an identical image, enlarged so that the lines (periodicity) could be seen. The span is about 9-10nm line to line, and the fiber arrangement is double that…. between 19-20nm. It appears that the protein(s) oligomerized here can come as an interconnected (maybe twisted) double strand.

Lower magnification image of the portion of a macrophage from which the inset to the right and above (white box) and the measurements of distance between fibrills (white lines)and smallest periodicity (blue box) measurements were made. Red dots=ribosomes at an estimated 27nm for reference; blue line = smallest periodicity; purple line = the 9-10nm pair, twisted, representing the fibrills (about 20nm in thickness if section is longitudinal, or diameter ir fibrill is cross sectioned. A measurement of about 20+nm is found from center of one fibrill, perpendicularly, to the center of the adjacent fibrill, (white connected lines on the bottom micrograph show distances between fibrils measured. Image at bottom has blue line of @20+nm –the approximate diameter of the “fibril”, and the dividing line of the two strands is visible.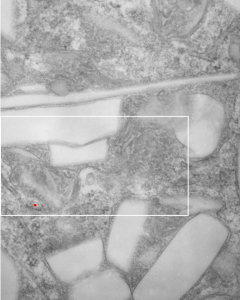
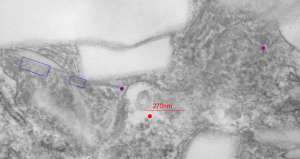
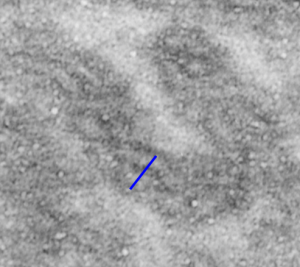
Perfluorodecyl iodide crystal periodicities
Perfluorodecyl iodide crystal periodicities are something that certainly looks like a phenomenon visible with electron microscopy which sampled such crystals after two hundred plus days residence in mouse macrophages. The crystals vary in size several orders of magnitude both in width and length, some appearing like little fractures of the whole. On the long ends of the IPFD crystals (within phagolysosomes, and surrounded by lysosomal enzymes which show their own type of periodicity (kind of a tubular or rounded structure — maybe both) protein strands trail out in a very distinct sweeping pattern. I have measured 15 distances between dense areas in this pattern (marked by black lines in the figure on the right and shown in array as white lines stacked on the left of that same micrograph). Taken back to the mean size of a ribosome (see image on left hand size – red dots are over ribosomes, and equate to 27nm diameter) the space between these strands coming from the long ends of IPFD crystals are about 10nm in width. I had sort of expected them to be closer to the 15nm measured in previous posts, but this is pretty close, and there is always variability from tangential sectioning and other artifacts.
On the long ends of the IPFD crystals (within phagolysosomes, and surrounded by lysosomal enzymes which show their own type of periodicity (kind of a tubular or rounded structure — maybe both) protein strands trail out in a very distinct sweeping pattern. I have measured 15 distances between dense areas in this pattern (marked by black lines in the figure on the right and shown in array as white lines stacked on the left of that same micrograph). Taken back to the mean size of a ribosome (see image on left hand size – red dots are over ribosomes, and equate to 27nm diameter) the space between these strands coming from the long ends of IPFD crystals are about 10nm in width. I had sort of expected them to be closer to the 15nm measured in previous posts, but this is pretty close, and there is always variability from tangential sectioning and other artifacts.
Speaking to artifacts…. the periodicity here cannot be attributed to some random precipitation of whatever-proteins…. since a clear and even density in background glumping of fixative and lose proteins around during fixation is seen in the “spaces” once occupied by perfluorodecyl iodide. Comparing with that background… this is pretty clear patterning.
Left hand image: IPFD inclusions in a macrophage taken from mouse liver where the animal had received IPFD 267 days prior to euthanasia (details in earlier posts). This is a small portion of a single macrophage and the light areas are the footprints of IPFD crystals. Most of this image, too, is within a single variably-shaped phagolysosome, filled with IPFD crystals and a curley but tubular shaped (oligomerized?) electron dense lysosomal protein(s). Middle images is magnified from box in left hand image, and right hand image is magnified from box in middle image. Middle and right hand images show the periodicity at IPFD crystal “ends”. I am trying to find any information on the crystalline structure of IPFD from the chemical databases… but have not found it yet.
Iodoperfluorocarbons
I found a very old article, portion below I did not retype but added the reference at the top, which mentioned briefly what I think is perfluorodecyl iodide (1-iodoperfluorodecane). I read this to mean that it is crystalline in nature and that the molecule has dimensions which might indicated that it has dimerized. If I knew what a “unit cell” was I might be able to relate it to the size (which is given here in Angstrom units) of 15.5 to 16, I might be able to figure out whether the periodicity found at the long ends of the IPFD crystals in vivo (within lysosomal structures) and the round organization seen in some images have relevance to that dimension.
 I found a more current article which seems to rehash some of what this early article by Rondesvedt mentioned in 1969. I like that he mentions that iodoperfluoroalkanes are bio-persistent, and that fits with the “mouse emulsion infusion” data that I see. That second publication is here. But there is no way without learning chemistry I can figure it out.
I found a more current article which seems to rehash some of what this early article by Rondesvedt mentioned in 1969. I like that he mentions that iodoperfluoroalkanes are bio-persistent, and that fits with the “mouse emulsion infusion” data that I see. That second publication is here. But there is no way without learning chemistry I can figure it out.
And in terms of environmental impact of PFC, in particular PFOS and PFOA and other perfluorochemicals in wide-spread usage, this is a nice article to read.
from which is a nice quote “Since the first reports revealing the widespread global occurrence of PFOS in wildlife (Giesy and Kannan 2001) and the frequent detection of PFASs in human blood (Hansen et al. 2001) were published a decade ago, the scientific literature on the environmental and toxicological aspects of PFASs has burgeoned rapidly, and the rate of publication currently exceeds 400 articles per year. In the existing body of literature, including governmental reports, authors have created terminology, names, and acronyms to describe these substances. Unfortunately, inconsistencies have inevitably arisen between various groups of authors. In the absence of any concerted effort between scientists to agree on a common terminology to designate the substances, a given compound has often been denoted by a variety of different names and acronyms, or a given acronym has been used to represent different substances. In addition, names to describe broad groups of substances have proliferated that in some instances mistakenly portray substances that are very different from one another as being the same. As a result, the scientific literature for these substances has at times become confusing. There is a need for harmonized terminology, names, and acronyms that clearly and specifically describe PFASs.” showing how difficult it is to read the literature on these chemicals.
I had to look up the words “unit cell” not being a chemist I had no clue what that meant… so here is a definition — A unit cell is the most basic and least volume consuming repeating structure of any solid. It is used to visually simplify the crystalline patterns solids arrange themselves in. When the unit cell repeats itself, the network is called a lattice.
I wonder why!
I wonder why i was “lucky/unlucky” enough to inherit the genes I did — jus t need to move all the time, perhaps to reinforce that I am “alive” — just need to think of stuff to do all the time, perhaps to keep myself “awake” — just need to be outside, in the sun, maybe to collect the sun’s “energy” — just need to think there is someone who will “catch” me when i fall, — just need to know the eternal, the unknowable, can it give me “peace”, — just “need” without “satiety” for 74 years.
t need to move all the time, perhaps to reinforce that I am “alive” — just need to think of stuff to do all the time, perhaps to keep myself “awake” — just need to be outside, in the sun, maybe to collect the sun’s “energy” — just need to think there is someone who will “catch” me when i fall, — just need to know the eternal, the unknowable, can it give me “peace”, — just “need” without “satiety” for 74 years.

