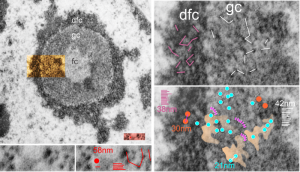
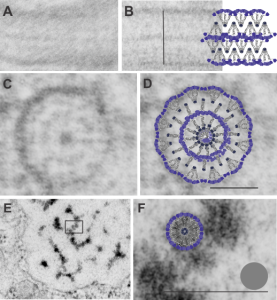
Shapes from biology (especially microscopy) sometimes are reminiscent of the most mundane objects in our daily lives. This nucleolus has reminded me of a donut with sprinkles for several decades. In searching the nucleolus for clues to the connection between the sizes of apparent granule-fiber, beads on a string, things to what has been reported in the molecular structures online, I used this particular nucleolus (from a baboon that had been given perfluorophenanthrene emulsion (probably IV)) and some tissues biopsied. This is lung. In other views, this particular cell (not clearly identifiable but likely an alveolar macrophage) contains PFC droplets.
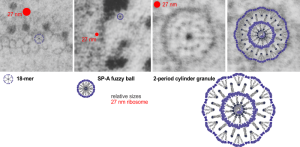
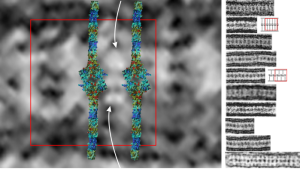
The “sprinkles” for this donut represent the dense fibrillar component (entities measured on a post from yesterday; the cake part of the donut is represented by the circular structure in the center of the side-by-side identical micrographs, which is the granular component. NB, the granular component on the left is light grey and round with the fibrillar center being the center of the donut. It was noted in some of these nucleolar structures that there is such definite banding and that the lumps on a string is a very good casual description of the organization of RNP along a template and the measurements are pretty good. As a reference point the diameter of a ribosome is about27 nm and the space between ribosomes in this particular cell is around 58 (or 2x the diameter of a ribosome). The diameter of the beads in the granular portion is 21nm, with a distance separating them of from adjacent structures is quite large and prominent, around 42nm (twice the diameter of the granule and though smaller in overall size than the ribosome and its space) is quite similar to it. The granules on a string in the dense fibrillar portion of the nucleolus is close to 30nm, slightly bigger than the average ribosome, and the space around these larger granules is the smallest among the three, at 38nm.
A second organization to the granular portion is seen in the parallel alignment and even spaceing of the granue. Orange lines lie over the very obvious linear pattern in the granular center of the nucleolus. A similar linear alignment is found in the dense fibrillar portion which becomes very very obvious in cajal bodies (not seen in this micrograph).
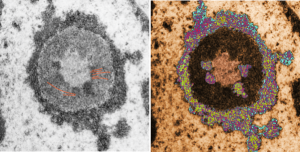 This image would be a fun cover submission… maybe someday i will make up a panel with three or four TEM look alikes. I know i have a valentine, and a ghost and a donut.
This image would be a fun cover submission… maybe someday i will make up a panel with three or four TEM look alikes. I know i have a valentine, and a ghost and a donut.