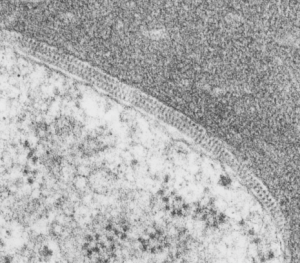
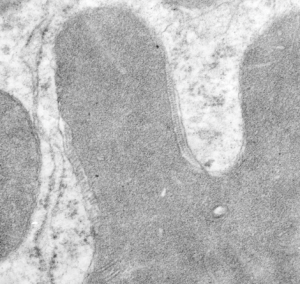
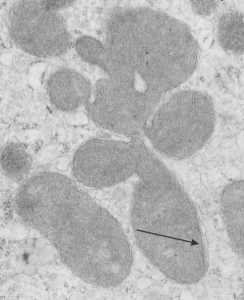
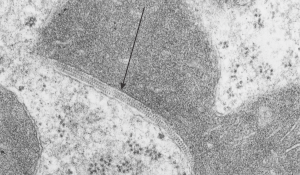
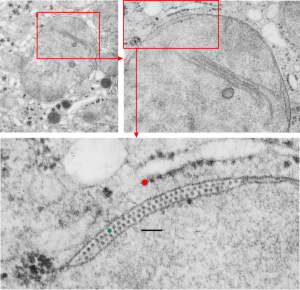
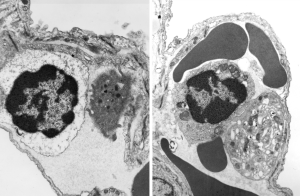
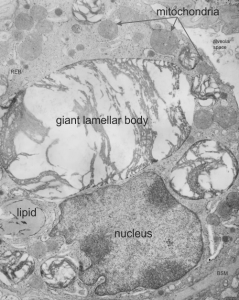
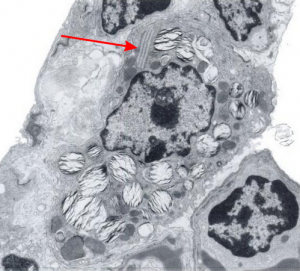
Quick comparison: vinyl chloride exposure vs toluene diisocyanate in lung — alveolar type II cells in guinea pig. Not going into much detail, but just an observation after comparing two strains of guinea pig (Hartley and English short hair (males)) from two different experimental protocols, each having their own age matched controls (from experiments performed way back in the 1980s by Dr. Martha Radike and Dr. Stuart Brooks (respectively) and looking just quickly at hundreds of images of type II cells (looking in fact for surfactant protein granules – an entirely different study) I would say that vinyl chloride exposure did two things quite differently than toluene diisocyanate. I am not going to dig up the exposure protocols, days exposure and stuff because it is published somewhere, but vinyl chloride definitely caused an increase in the lamellar body size, and a concommitant decrease in the amount of RER and other organelles in the cytoplasm. Lamellar bodies got very large, caused indentations to nuclei and sometimes fused into these really large lumpy lamellar bodies. In addition mitochondria got large, had cristae deformed by some kind of matrix issue, the electron density was not decreased in the matrix, but cristae looked pushed and squeezed, in addition to an occasional fluting of the outer mitochondria membrane. (BTW, liver mitochondria from vinyl chloride exposed animals did show differences from unexposed as well). Figure from neg 6476, block M8005, anm# 51, vinyl chloride, low vitamin C. See below, two great nucleoli, several mitochondria with diminished cristae to mitochondrial ratio, enlarged matrix, and odd shapes within mitochondria. The RER in the cytoplasm doesnt fig the granular layered profiles that have been seen in guinea pig that I am trying to establish as surfactant protein A, but are dilated and atypical for an alveolar type II cell from an untreated, and well, guinea pig.
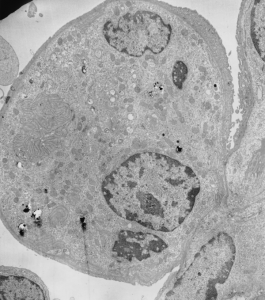
Toluene diisocyanate on the other hand (and some of the control animals along with… so you can judge whether this is an animal room sickness affecting all guinea pigs or something unknown) did NOT show mitochondrial or lamellar body size increase.