Eclipse through xray film, pinhole on my mini-cooper (waxing), through a strainer on a rock walkway (waning), and pinhole on white paper.
Monthly Archives: August 2017
This is the disparity that makes me grieve
50 ways to name your compound: PFOB
Paul Simon’s “50 ways to leave your lover” was an hysterically clever and popular song
“Just slip out the back, Jack
make a new plan, Stan
Don’t need to be coy, Roy
just listen to me
Hop on the bus, Gus
don’t need to discuss much
Just drop off the key, Lee
and get yourself free,…”
Of these lyrics I am reminded, this morning, with 50 ways to name the same compound, PFOB. Yep really there are almost 50 in this list according to PubChem (similarly for ChemSpider) for perfluorooctylbromide, which back in the 1975s we nicknamed PFOB (yep PFOB is still on the list). There is something wrong with the system when there are 50 ways to name the same thing (apparently this is the same compound, though I seriously doubt it. What we called PFOB was the neat liquid, others may have add things??, but consider that we don’t even have 50 names for LOVE, why should there be 50 names for this simple perfluorinated molecule. Such naming schemes more than doubles the amount of work necessary to figure out how to read research papers that use PFOB.
It was reported several places in the literature (particularly as LiquiVent (Allied Pharmaceutical Corporation San Diego California)) and it seems like they had a big hand in making up other names for it as well.
Compound names used for PFOB (ok there are only 49, not 50)
CAS No. 423-55-2 (Chemical Abstracts Service)
1-Brom-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluoroctan
1-Bromo-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluorooctane
1-Bromo-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadécafluorooctane
1-Bromoheptadecafluorooctane
1-Bromoperfluorooctane
bromoperfluorooctane
bromure de n-perfluorooctyle
Foralkyl Br 8
Heptadecafluoro-n-octyl Bromide
Heptadecafluorooctyl bromide
n-Perfluorooctyl Bromide
Octane, 1-bromo-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluoro-
Perflubron
perfluoro(octyl) bromide
Perfluoroctyl bromide
perfluoroctylbromide
perfluorooctyl bromide
perfluorooctylbromide
1-bromo-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluoro-octane
1-Bromoperfluorooctane,
1-Bromoperfluorooctane; 1-Bromo-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluorooctane
2016022
207-028-4
bromure de n-perfluorooctyle
CHEBI:38803
D05439
EINECS 207-028-4
IMAGENT
Imagent (TN)
Imagent BP
Imagent GI
LiquiVent
Long’s compound
NCGC00166217-01
Octane, 1-bromoheptadecafluoro-
Oxygent
oxygent ca
Oxygent HT
Perflubron (USP)
perflubron emulsion
Perfluoro-n-Octyl Bromide
perfluoro-octylbromide
PFOB
Q1D0Q7R4D9
QA-3920
RG8700000
UNII:Q1D0Q7R4D9
UNII-Q1D0Q7R4D9
Inert and partially inert particles in lysosomes
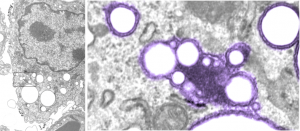
Here is something that might be close to looking at the endosome-to-lysosome pathway taken by perfluorochemical droplets in experiments either of liquid breathing or infusion of perfluorochemical-based blood substitutes. I this particular article (Zhou et al, Int J. Nanomedicine, 2010 — might be a good journal to submit the PFC papers upcoming). It is nice to see that the endosomes look a little like the dense, highly enzyme filled, MVB/LE and lysosomes seen in macrophages and other inclusions in cells exposed to PFC. I am excerpting and editing one of their TEMs… without permission, but giving them credit, to make this parallel. Particles (Poly(d, l-lactide) (PLA) and poly-d, l-lactide-poly(ethylene glycol) (PELA) with PEG weight ratios of 10%, 20% 30%? I think this is all) don’t show a two phase morphology with their SEM images, but clearly with TEM there is a dense core and a lighter outer area. This is in total contrast to the “footprint” of PFC, in the image on the left which is basically “completely electron lucent”. But there are some similarities in the lysosome (called that just for simplicity) in each of the two images, though the PFC lysosomes (on the left) really are densely packed with enzymes, maybe even more than the nanoparticles (on the right). SEM of nanoparticles, see inset on the right.
Lysosomes, diagrams, hysterical typo
 I hope I am able to laugh at my mistakes, as this individual needs to laugh at their typo. What… ha ha… a lysosomal rapture…. no kidding, waiting for the rapture myself, and hoping to be among those taken into the ether. Just in case you missed it…. lysosomal rupture would be, i must presume, what was intended.
I hope I am able to laugh at my mistakes, as this individual needs to laugh at their typo. What… ha ha… a lysosomal rapture…. no kidding, waiting for the rapture myself, and hoping to be among those taken into the ether. Just in case you missed it…. lysosomal rupture would be, i must presume, what was intended.
Perhaps this book chapter is source of the “rapture”
Some people just don’t get it
OK mr RotoRooter, whomever you are on Red Bud Avenue 45229 8/17/2017 at 9:40 in the morning. When you have your windows up, sitting in your nice truck with ohio license plate PJA 1555, talking on your phone (very likely not related to company work), using up unnecessary fuel (which of course the customer will pay for) — do you not understand that YOU are part of the energy waste in this world, do you not understand that we CONSERVE energy because it is the the civil, earth-friendly, responsible way to behave. YOU are not the only one in the universe.
When I ask you to turn off your engine, and you scowl at me for mentioning your carbon footprint, conservation, the atmosphere, and that the customer pays for your sitting in airconditioning talking on company time. No hope at all.
Perfluorocarbon compounds and lysosomes: slippery mix
I posted a few days ago (here) a phrase which i thought was a little off color, and definitely made for effect, but stirred up something of interest in the way that perfluorochemicals “slippery and inert” might create or produce unusual lysosomal structures. I did post a composit lysosomal structure which looked to be like a little pouch full of varying size beads, or droplets, which are presumed to be the footprints of perfluorochemical left after liquid breathing or invusion of PFC emulsions. I examined some additional lysosomes this morning and it is clear there are many tiny droplets that are mixed in among some very large droplets, and these are so close to the nucleus, and near, perhaps almost looking like a connection, some of the ER in the cell which could be classified as RER. PFC ispossibly moving back up the ER profiles, and while all the names for endosomes (early, late, hybrid, budding, bla bla) this system is obviously continuous and naming “early” “late” etc adds little to the understanding (in my humble opinion), but naming them by pH and activities and/or membrane proteins associated, would be beneficial. Another issue is why there is a distinct dark band around the PFC droplets, it gives the appearance of TWO trilaminar membranes rather than a typical membrane bound endosome or lysosome.
Below is a micrograph (actually a scan of the original negative since the print I had marked and draw all over) which has two magnifications. Top image has white box which is enlarged below it, and the long stringy lysosomal structures are very close to the nucleus, even adjacent RER. this is almost like the “egg in snake” appearance. Presumptive perfluorochemical droplets (in this case E2 — structure given HERE) are colored green, the most electron dense areas surrounding the green (darker green) are lysosomal enzymes (also presumptive since I have not done any specific staining to verity it… but it is obvious.) White arrows point to the tiniest droplets (some in the range of about 30 nm, and some of those light areas may be tangential cuts off the ends of larger particles.
I suppose it is possible that the movement of PFC up the RER is to be expected because of its spreading chemical properties (at least according to Jean G. Riess and Marie Pierre Krafft.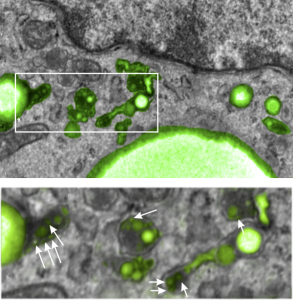
Love casts out fear and hate
 “No one is born hating another person because of the color of his skin or his background or his religion … People must learn to hate, and if they can learn to hate, they can be taught to love … For love comes more naturally to the human heart than its opposite,” Obama said, quoting former South African president Nelson Mandela”. This is lifted from a google search for Obama’s tweets about Charlottesville violence.
“No one is born hating another person because of the color of his skin or his background or his religion … People must learn to hate, and if they can learn to hate, they can be taught to love … For love comes more naturally to the human heart than its opposite,” Obama said, quoting former South African president Nelson Mandela”. This is lifted from a google search for Obama’s tweets about Charlottesville violence.
It brought tears to my eyes and reinforced the differences in style between this past president and the current one. The difference is as big as the grand canyon.
But i also am a biologist, and also a student of religion. It is clear that identification of “self” and “others similar to self” is a big part of survival, procreation, continuation of the species. We need to be taught to “hate” but we are wired to “love like minded and like-looking humans”.
Some religions, cultures, philosophies, teach that LOVE casts out fear, and it is typically fear that causes us to “hate” or be “aggressive”. Other religions teach people to “HATE and OBLITERATE” what is different in an effort to allay their fears about survival. Fear itself is either learned, or experienced or both. Extrapolate teachings and experiences, to unhealthy mindsets, and daily situations, and one ends up an enormous amount of fear, hate and violence. We need to overcome fear with the more powerful force, which is LOVE.
Not typical lysosomes: Do engulfed PFC droplets migrate up the ER
Looking at lysosomes in the lung of mice which have breathed perfluorochemical (PFC) liquids is interesting. These are old archived micrographs from the early 1970s but have a lot of valuable information about the reactivity of perfluorochemicals in the body (the latter being touted as totally inert. Regardless of what science says about the reactivity of PFC they do cause a mountain of effects in humans, not the least of which is an immediate uptake by the reticuloendothelial system, massive sequestering and dumping of enzymes into the resultant lysosomes, and a kind of partitioning, or re-partitioning, or re-emulsification of the PFC, depending upon whether it was breated as a neat liquid, or infused as an emulsion with other agents accompanying it.
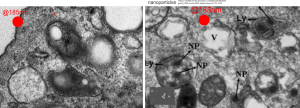
So “case in point” is this alveolar macrophage, picture taken from the lung of a mouse which breathed E2 for 3 hours and was allowed to recover for 48 hours then lungs taken for electron microscopy (and probably chemical analysis, though I have no clue where those data are at this point 40 years down the pike). I just know that my negative number is 1351, and that block number is 4840, magnification was 5,000 (Zeiss 10) date 2 17 1975 at 60kv. Mouse weighed 16.85g, breathed E2 for 3 hours on 2 3 1975, and was processed on 2 5 1975, which equates to 48 hour recovery. The sample of lung was taken from the R, mid lobe.
The image below (L) is a portion of the original negative (scanned as a transparency from the film) with an enlargement (box demarcates it) on the right where I have “pseudocolored” the protein densities with purple, which surround the empty footprints where the PFC was fixed (leaving the nice round spaces) in the cytoplasm. About 6 ribosomes (@27nm diameter) are also highlighted beneath the central phagolysosome?lysosome?membrane structure to show that the size of the smallest perfluorochemical droplets is in the nanometer range, before — before what, perhaps being small enough to diffuse back into the alveolar air space and be exhaled. I don’t believe anyone knows whether the larger or the smaller E2 droplets are more apt to move into the alveolar air space. There is one tiny droplet which is about 27 nm nesteled between two ribosomes… go figure. The enzyme response here is pretty amazing, and appears in section to be most often seen as spheres, but is more likely to be a longer sausage shaped lumpy body.
The bottom line of this image is that these droplets appear to migrate along the membrane systems.
Some perfluorocarbon based blood substitute – “trivia” or not so trivial
The febrile reaction to PFC based blood substitutes was purported (Donat R Spahn, Blood substitutes Artificial oxygen carriers: perfluorocarbon emulsions. Crit Care. 1999; 3(5): R93–R97) to be related to particle size of the emulsified infusion fluid. Lower particle size was stated to reduce the uptake by the reticuloendothelial system, which increased tolerability, as that stimulation of phagocytosis, and what must be (unmentioned) feedback loops, was deemed responsible for fever and flu like symptoms in patients receiving the blood substitues.
Spahn quotes statistics for IV administration of Oxygent “Intravascular half life is dose-dependent, and for Oxygent was found to be 9.4 ± 2.2 h for a dose of 1.8 g/kg [4]. After the initial uptake of the perfluorocarbon emulsion into the RES the droplets are slowly broken down, and the perfluorocarbon molecules are taken up in the blood again (bound to blood lipids) and transported to the lungs, where the unaltered perfluorocarbon molecules are finally excreted via exhalation. At present no metabolism of fluorocarbon molecules is known in humans.”
I didn’t see any micrographs which gave clue as to “break-down” of droplets, so this is confusing. I have seen completely solid “footprints” of fluorocarbons, round and with lysosomal enzymes, sometimes a lot, surrounding or off to one side of a droplet, I have also seen the frothy huge lysosomal inclusions which do not have the orderly rounded appearance.